The GansCoater® - The World's #1 Automated Tablet Coating Machine for Regulated Pharmaceutical Manufacturing
Built on more than 50 years of coating expertise, the GansCoater® is the highest-selling automated tablet coating machine to US-FDA, UK-MHRA, and EMA regulated companies globally. With over 2000 active installations across 50+ countries and 40+ models covering batch sizes from 40 grams to 1.2 tonne, the GansCoater® delivers consistent, reproducible coating results at every scale of production.
Validated by IIT Mumbai
What makes the GansCoater® different
Aeroflo® patented baffle system
- Spray losses below 5% - less suspension waste, lower operating cost per batch
- Zero dead zones - uniform tablet exposure to spray and drying air throughout the pan
- Tablet slippage eliminated - consistent tumbling motion regardless of batch load
- 30% less compressed air - compared to industry standard baffle designs
- A higher turndown ratio than the standard specified capacities can be achieved by installing optional “reduced baffles” upon request.


Aeroflo® patented baffle system
- Spray losses below 5% - less suspension waste, lower operating cost per batch
- Zero dead zones - uniform tablet exposure to spray and drying air throughout the pan
- Tablet slippage eliminated - consistent tumbling motion regardless of batch load
- 30% less compressed air - compared to industry standard baffle designs
- A higher turndown ratio than the standard specified capacities can be achieved by installing optional “reduced baffles” upon request.

Interchangeable pan system
- R&D to commercial scale on one platform - 40g through 1200kg
- Six-minute single operator changeover - validated, tool-less pan exchange
- Large turndown ratio - flexible processing within each pan size
Precision spray and drying system
The GansCoater® spray system is engineered for uniform droplet size, wide spray coverage, and zero tablet bearding. The directional air plenum guides drying laminar air through the tablet bed for maximum drying efficiency. Non-contact infrared sensors measure tablet bed temperature continuously, providing real-time data without contamination risk.
- Anti-bearding spray nozzles - no elastomeric contact parts, easy maintenance, lower risk
- Infrared tablet bed thermometry - 10–300ms response time, non-contact measurement
- Directional air plenum - maximises drying efficiency per unit of energy input
- Ultrasonic nozzle-to-bed distance sensors can be integrated to monitor, validate, and optimise coating suspension utilisation while minimising spray drying losses.


Precision spray and drying system
The GansCoater® spray system is engineered for uniform droplet size, wide spray coverage, and zero tablet bearding. The directional air plenum guides drying laminar air through the tablet bed for maximum drying efficiency. Non-contact infrared sensors measure tablet bed temperature continuously, providing real-time data without contamination risk.
- Anti-bearding spray nozzles - no elastomeric contact parts, easy maintenance, lower risk
- Infrared tablet bed thermometry - 10–300ms response time, non-contact measurement
- Directional air plenum - maximises drying efficiency per unit of energy input
- Ultrasonic nozzle-to-bed distance sensors can be integrated to monitor, validate, and optimise coating suspension utilisation while minimising spray drying losses.
Containment for potent compounds
The GansCoater® is available in OEB Level 5 containment configurations for the processing of highly potent active pharmaceutical ingredients (HPAPIs).
- Contained designs include fully closed charging, in-process sampling, and discharging systems - eliminating operator exposure at the highest-risk stages of the coating process.
- Isolation technology is integrated directly into the coating system architecture, not retrofitted.
- BIBO dust collector with HEPA H14 filter - DOP tested, DP monitored
- Full-face screening probes continuously monitor air from the inlet and/or exhaust HEPA filters, detecting any filter integrity breach and automatically initiating safety measures to isolate and protect the equipment, product, and process.

Automation and Control
The GansCoater® automation platform is purpose built for pharmaceutical regulatory compliance – not adapted from a generic industrial control system. The system delivers:
- 21-CFR part 11 compliance for data integrity and storage, ALCOA+ compliant
- ALCOA+ data integrity - attributable, legible, contemporaneous, original, accurate and complete records
- Recipe driven operation - parameter-locked process control with role-based access
- IoT connectivity - pre-emptive analytics, remote monitoring, condition-based maintenance alerts
- Pharma 4.0 ready - MES/ERP integration via OPC-UA, real-time process dashboards
- GAMP 5 validation - computerised system validation documentation supplied as standard
Installed in facilities regulated by US-FDA, UK-MHRA, EMA, WHO-GMP, Japan-PMDA, TGA, SAHPRA, MCC, and health authorities in 40+ countries. CE, UL, ATEX, ISO, and IEC configurations available on request.
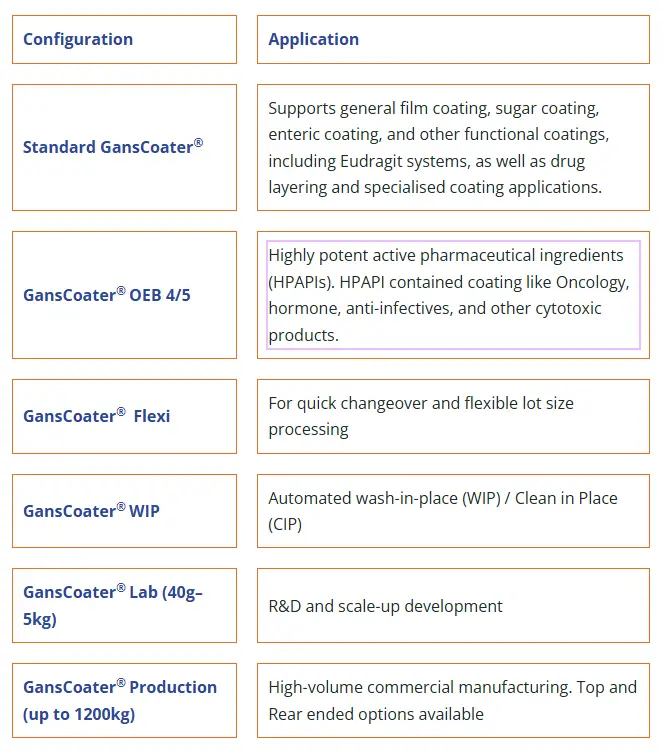
Available configurations
Configuration
Application
Supports general film coating, sugar coating, enteric coating, and other functional coatings, including Eudragit systems, as well as drug layering and specialised coating applications.
GansCoater® OEB 4/5
Highly potent active pharmaceutical ingredients (HPAPIs). HPAPI contained coating like Oncology, hormone, anti-infectives, and other cytotoxic products.
GansCoater® Flexi
For quick changeover and flexible lot size processing
GansCoater® WIP
Automated wash-in-place (WIP) / Clean in Place (CIP)
GansCoater® Lab (40g–5kg)
R&D and scale-up development
GansCoater® Production (up to 1200kg)
High-volume commercial manufacturing. Top and Rear ended options available
Available configurations

Feasibility Batch
40 grams to 400 grams

R&D
400 grams to 5 Kg

Pilot Scale
7 Kg to 75 Kg

Commercial Scale
75 Kg to 400 Kg

Bulk Production
450 Kg to 1200 Kg
Frequently asked questions -GansCoater®
What batch sizes does the GansCoater® handle?
Is the GansCoater® compliant with FDA and cGMP requirements?
What types of tablet coating can the GansCoater® perform?
The GansCoater® supports film coating, enteric coating, functional multi-layer coating, sugar coating, and active drug layering. The system is optimised for aqueous and solvent-based coating formulations across all solid dosage form types including tablets, capsules, minitablets and other solid forms >2mm size.
Does the GansCoater® support HPAPI and OEB Level 5 processing?
Yes. Gansons offers OEB Level 5 containment configurations for the GansCoater®, including fully closed charging, in-process sampling, and discharging systems. These designs protect operators from exposure to highly potent active pharmaceutical ingredients (HPAPIs) at every stage of the coating cycle.
What is the Aeroflo® baffle system?
How does the GansCoater® support Pharma 4.0?
The GansCoater® automation platform is IoT-enabled and supports Pharma 4.0 with real-time process monitoring dashboards, recipe-driven operation with parameter locking, electronic batch records, and remote diagnostic capability. Pre-emptive IoT analytics monitor equipment health continuously, enabling condition-based maintenance that prevents unplanned downtime.
What documentation does Gansons provide for coating machine qualification?
Gansons provides FAT (Factory Acceptance Testing), SAT (Site Acceptance Testing), IQ (Installation Qualification), and OQ (Operational Qualification) documentation support as standard. GAMP 5-compliant computerised system validation documentation is included with the automation package. Material traceability certificates and calibration records are supplied at equipment handover.
How many GansCoater® units have been installed globally?
Over 2,000 GansCoater®
units are installed globally, making it the highest-selling automated tablet coating machine to regulated pharmaceutical facilities worldwide. Installations span 50+ countries including the United States, European Union, United Kingdom, India, Africa, Southeast Asia, and Latin America.

Quality, certifications & compliance
- ISO 9001:2015: certified manufacturing across 250,000+ sq ft
- cGMP design: 85%+ of installations in US-FDA, UK-MHRA & EMA regulated facilities
- 21 CFR Part 11 / EU Annex 11: data integrity and audit trail built in
- GAMP 5: computerised system validation documentation supplied
- ATEX / IECEx: solvent and HPAPI configurations available
- CE marking: full technical file for EU supply
- FAT, IQ/OQ : qualification documentation as standard
Compliance and Certifications






Downloads
- Brochure - GansCoater®
- Case study - GHPN III
- Research Paper - By IIT, Bombay
Connecting Globally, Serving Locally
With offices in Mumbai, India, and London, UK, Gansons operates globally while upholding a strong local commitment. Our extensive international network of partners and agents ensures personalized support for customers in 50+ countries where they operate.
We understand the critical importance of uninterrupted production. That’s why we’ve developed a robust spare parts program that ensures your production never falters. Moreover, our global network of trusted subvendors and automation providers stand by your side, providing the necessary support to keep your operations running smoothly.

