Ointment Preparation Plant
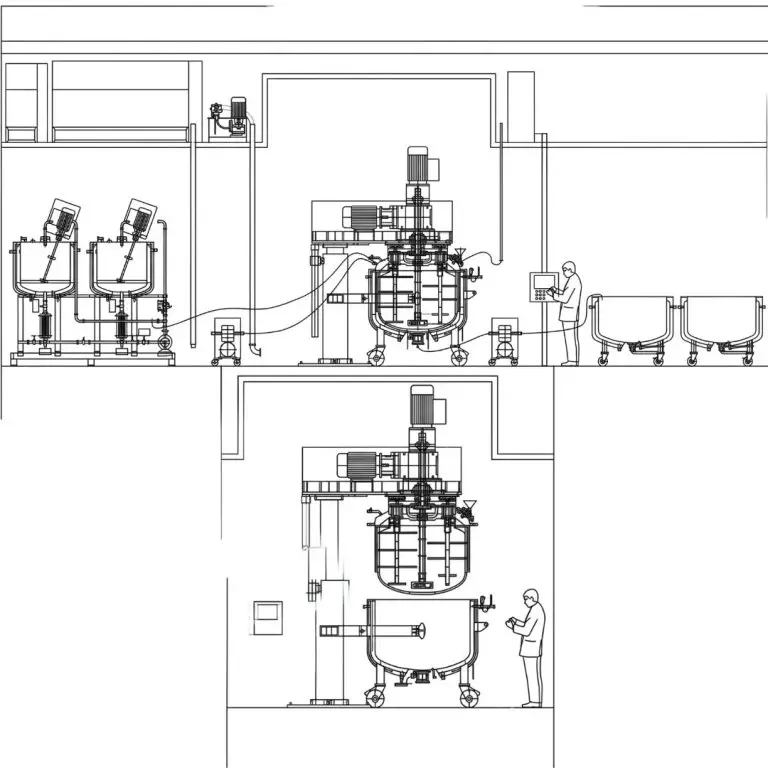
The Ointment Manufacturing Plant by Gansons is meticulously engineered to serve the pharmaceutical and cosmetic industries, supporting the production of a wide range of products, including ointments, creams, lotions, and toothpaste. The facility is designed with a strong emphasis on minimizing manual handling and maintaining consistent vacuum levels during mixing and blending, ensuring superior product quality.
Operational Efficiency
- Closed-Circuit Processing: Wax and water phases are heated and vacuum-transferred through a conical filter into the manufacturing vessel to ensure a sterile, contained environment.
- Automated Emulsification & Transfer: An integrated Ointment Transfer Pump handles both product recirculation during emulsion and final discharge into the storage vessel.
- Optimized Resource Management: The streamlined system allows for single-operator control, significantly improving manpower utilization and lowering operational costs.
Compliance and Quality Assurance
The Gansons Ointment Manufacturing Plant adheres to Current Good Manufacturing Practices (cGMP), featuring paint-free construction and high-quality materials. All contact parts are made of stainless steel (SS 304), with SS 316 available upon request. The plant includes features such as:
- Silicone (Food Grade) Gaskets: Ensuring safety and compliance.
- High-Speed Emulsifiers: For effective mixing.
- CIP & SIP Connections: For cleaning and sterilization.
- Digital Monitoring: For temperature and time settings, providing real-time data to operators.


Compliance and Quality Assurance
The Gansons Ointment Manufacturing Plant adheres to Current Good Manufacturing Practices (cGMP), featuring paint-free construction and high-quality materials. All contact parts are made of stainless steel (SS 304), with SS 316 available upon request. The plant includes features such as:
- Silicone (Food Grade) Gaskets: Ensuring safety and compliance.
- High-Speed Emulsifiers: For effective mixing.
- CIP & SIP Connections: For cleaning and sterilization.
- Digital Monitoring: For temperature and time settings, providing real-time data to operators.
The Process
The Gansons Ointment Manufacturing Plant operates as a closed circuit manufacturing facility, ensuring efficient and hygienic production of ointments and creams. Below is a structured breakdown of the system’s key components and processes.

Feeding of Wax/Water Phase
- Materials Used: Wax and water.
- Temperature Control: Both wax and water are heated to the required temperature before transfer.

Transfer to Manufacturing Vessel
- Method: The heated wax and water are transferred to the Manufacturing Vessel or Planetary Mixer.
- Vacuum Transfer: This transfer is achieved through a vacuum system using a conical filter, ensuring contamination-free movement of materials.

Storage Vessel Operations
- Fixed Position: The Storage Vessel remains fixed in place after filling.
- Connection to Filling Area: It is connected to the Ointment Transfer Pump for further processing.
- Transfer to Filling Machine: The pump transfers the ointment from the Storage Vessel to the hopper of the Ointment Filling Machine.

Emulsion Formation
- Recirculation: During the emulsion formation process, the product is recirculated through the Ointment Transfer Pump.
- Discharge: The pump also discharges the emulsified product into the Storage Vessel.

Automation and Control
Gansons equipment is available with an integrated intelligent automation platform. Key features include:
- PLC/SCADA-based control with touch-screen HMI
- Centralised recipe management with role-based access control
- IoT enabled for remote monitoring and pre-emptive analytics
- Integrable with any site architecture including MES, ERP, and LIMS
- Customised reporting and smart graphs for lot reproducibility
- Industry-leading IPC processors for fast, reliable control performance
- 21 CFR Part 11 compliant automation available as an add-on for regulated pharmaceutical facilities
- GAMP 5-aligned computerised system validation documentation supplied with 21 CFR Part 11 configurations
Installed in facilities regulated by US-FDA, UK-MHRA, EMA, WHO-GMP, Japan-PMDA, TGA, SAHPRA, MCC, and health authorities in 40+ countries. CE, UL, ATEX, ISO, and IEC configurations available on request.
Applications

Cosmetic Products
Face creams, sunscreens, and body scrubs.

Skincare Products
Creams, lotions, and ointments.

Hair Care Products
Shampoos, conditioners, and treatments.

Pharmaceutical Products
Antiseptics, anti-fungal ointments, and gels.
Capacity Range
10L to 1500L
Compliance and Certifications






Downloads
- Brochure – Ointment Preparation plant

